Oxidative Phosphorylation Summary
|
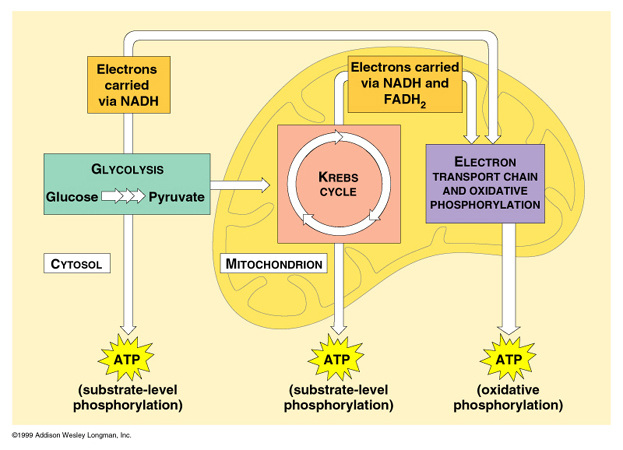
Location
Eukaryotes - inner membrane of mitochondria Prokaryotes- plasma membrane |
Inputs
NADH FADH2 O2 ADP Pi |
Outputs
NAD+ FAD H2O ATP |
Important Steps
All |
ATP produced per Glucose: 32-34
ATP produced per Acetyl-CoA: 11
ATP produced per Acetyl-CoA: 11
Oxidative phosphorylation is the major aerobic process to generate ATPs for cellular energy. This occurs in mitochondria of eukaryotic cells, specifically in the inner membrane and the intermembrane space. in prokaryotes, it occurs on the plasma membrane.
The process of Oxidative Phosphorylation is best explained by the chemiosmotic theory proposed by Peter Mitchell.
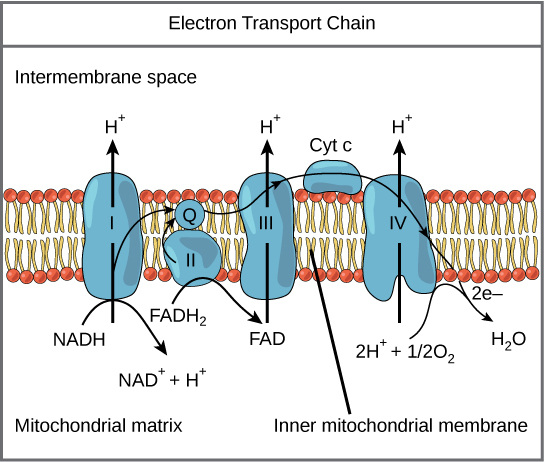
Electron Transport Chain Explained
WHEN: Following glycolysis and the citric acid cycle, NADH and FADH2 account for most of the energy extracted from food
"WHO": Most of the chain’s components are proteins, which exist in multiprotein complexes
The carriers alternate reduced and oxidized states as they accept and donate electrons
WHAT: These two electron carriers (NADH and FADH2) donate electrons to the electron transport chain, which powers ATP synthesis via oxidative phosphorylation
Electrons drop in free energy as they go down the chain and are finally passed to O2, forming H2O
Electrons are transferred from NADH or FADH2 to the electron transport chain
Electrons are passed through a number of proteins including cytochromes (each with an iron atom) to O2
The electron transport chain generates no ATP
WHERE: The electron transport chain is in the cristae of the mitochondrion
WHY: The chain’s function is to break the large free-energy drop from food to O2 into smaller steps that release energy in manageable amounts
Chemiosmosis Explained
the use of energy in a H+ gradient to drive cellular work
Electron transfer in the electron transport chain causes proteins to pump H+ from the mitochondrial matrix to the intermembrane space
H+ then moves back across the membrane, passing through channels in ATP synthase
ATP synthase uses the exergonic flow of H+ to drive phosphorylation of ATP
The energy stored in a H+ gradient across a membrane couples the redox reactions of the electron transport chain to ATP synthesis
The H+ gradient is referred to as a proton-motive force, emphasizing its capacity to do work
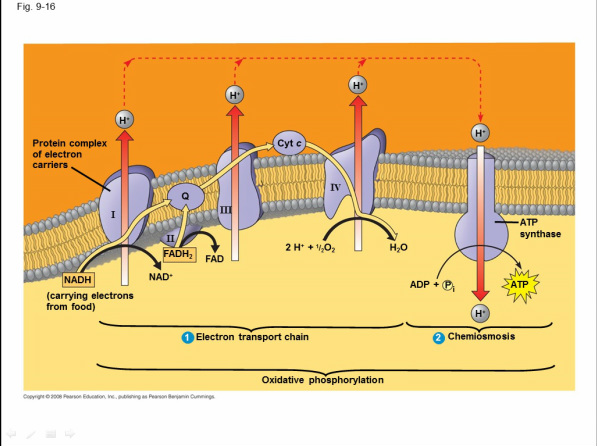
Electron Transport Chain Explained
WHEN: Following glycolysis and the citric acid cycle, NADH and FADH2 account for most of the energy extracted from food
"WHO": Most of the chain’s components are proteins, which exist in multiprotein complexes
The carriers alternate reduced and oxidized states as they accept and donate electrons
WHAT: These two electron carriers (NADH and FADH2) donate electrons to the electron transport chain, which powers ATP synthesis via oxidative phosphorylation
Electrons drop in free energy as they go down the chain and are finally passed to O2, forming H2O
Electrons are transferred from NADH or FADH2 to the electron transport chain
Electrons are passed through a number of proteins including cytochromes (each with an iron atom) to O2
The electron transport chain generates no ATP
WHERE: The electron transport chain is in the cristae of the mitochondrion
WHY: The chain’s function is to break the large free-energy drop from food to O2 into smaller steps that release energy in manageable amounts
Chemiosmosis Explained
the use of energy in a H+ gradient to drive cellular work
Electron transfer in the electron transport chain causes proteins to pump H+ from the mitochondrial matrix to the intermembrane space
H+ then moves back across the membrane, passing through channels in ATP synthase
ATP synthase uses the exergonic flow of H+ to drive phosphorylation of ATP
The energy stored in a H+ gradient across a membrane couples the redox reactions of the electron transport chain to ATP synthesis
The H+ gradient is referred to as a proton-motive force, emphasizing its capacity to do work
Here is a two part video to explain further.
Sathasivan, Kanagabapathi. Cell & Molecular Biology, An introduction 2nd Edition. Dubuque : Kendall Hunt Publishing Company, 2013 . Print.