BASIC REACTIONS OF AEROBIC RESPIRATION
4 Major Processes:
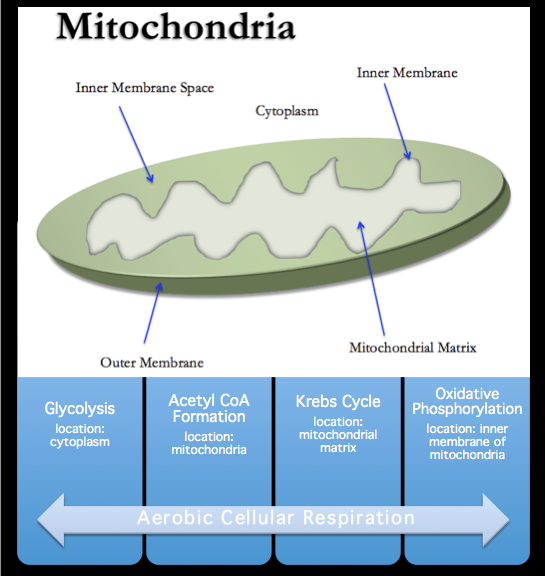
- Glycolysis takes place in the cytoplasm and is the first step to both aerobic and anaerobic respiration. It is the process that turns glucose into pyruvate and 2 ATP. The goal of this process is simply just the first step of releasing energy.
- Acetyl CoA formation: Once glycolysis produces the pyruvate, the pyruvate is transported inside the mitochondria and is then converted into Acetyl Co-A, where it proceeds into the Krebs Cycle.
- The Krebs Cycle, also known as the 'Citric Acid Cycle' occurs in the mitochondria's matrix. The Krebs Cycle works by taking its reactant from glycolysis, Acetyl Co-A, and produces energy carriers, 2 ATP, NADH, FADH2, and it's waste product, CO-2.
- Oxidative Phosphorylation: Electrons from NADH and FADH2 are then transferred to molecular oxygen, coupled to the formation of an additional 32 to 34 ATP molecules by oxidative phosphorylation. Electron transport and oxidative phosphorylation are critical activities of protein complexes in the inner mitochondrial membrane, which ultimately serve as the major source of cellular energy.
This table illustrates a simple interpretation of the order of reactions in aerobic cellular respiration and the location in which they occur.
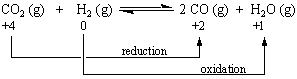
Redox Reactions
Redox reactions are a family of reactions that are concerned with the transfer of electrons between species. Redox reactions are a matched set -- you don't have an oxidation reaction without a reduction reaction happening at the same time. Oxidation refers to the loss of electrons, while reduction refers to the gain of electrons. You can remember oxidation and reduction with a simple acronym: OIL RIG (Oxidation Is Loss, Reduction Is Gain)
Why would you call something reduced if it is actually gaining an electron?
The answer lies in the charge of the atom. Since electrons are negatively charged, gaining an electron also means gaining a -1 charge, reducing the overall charge of the atom. Take a look at the example below to get a better understanding.
Why would you call something reduced if it is actually gaining an electron?
The answer lies in the charge of the atom. Since electrons are negatively charged, gaining an electron also means gaining a -1 charge, reducing the overall charge of the atom. Take a look at the example below to get a better understanding.
In cellular respiration, glucose is oxidized in a series of steps that release energy, little by little. The electrons that glucose loses as it is oxidized are usually picked up by NAD+, a coenzyme that acts as an electron carrier. In redox terms, NAD+ oxidizes glucose, and glucose reduces NAD+. When it is reduced, NAD+ becomes NADH because it gets an extra hydrogen atom and an extra electron. One proton goes off by itself (H+). During oxidative phosphorylation, the last electron acceptor is called the terminal electron acceptor. In organisms that breathe in oxygen, like us humans or animals etc., oxygen is the terminal electron acceptor. It is the last molecule to accept electrons and is therefore reduced.
Electron Carriers
NAD+ and FAD carry electrons to the electron transport system. In the electron transport system, NADH and FADH2 are used to produce ATP as electrons are passed from one carrier to another. Eventually the electrons combine with hydrogen ions and oxygen (reduction) to form water. Take some time to understand this concept by watching this short video clip.
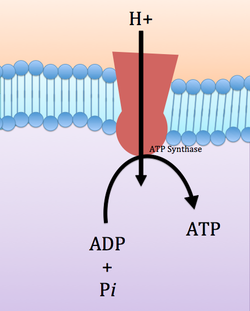
ATP Synthesis
|
ATP is synthesized by ATP synthase, a large complex of membrane bound proteins. The energy associated with they hydrogen proton gradient is used to synthesize ATP from ADP and Pi. This occurs at the ATP synthase complex. One hydrogen ion enters the ATP synthase complex from the intermembrane space and a second hydrogen ion leaves it on the matrix space. The upper part of the ATP synthase complex rotates when a new hydrogen ion enters. Once three protons have entered the matrix space, there is enough energy in the ATP synthase complex to synthesize one ATP. In this way, the energy in the hydrogen ion gradient is used to make ATP. Without a gradient, there is not more energy to make ATP. In biological systems however, the gradient is always maintained.
|
Terms to Know
Cellular Respiration: the set of the metabolic reactions and processes that take place in the cells of organisms to convert biochemical energy from nutrients into adenosine triphosphate (ATP), and then release waste products.Aerobic Respiration: a type of cellular respiration defined as the energy metabolism in the presence of oxygen.
Oxidation-Reduction (redox) reactions: the gain or loss of electrons. In a reduction reaction, electrons are gained. In an oxidation reaction, electrons are lost. Use the acronym OIL RIG to remember this :Oxidation Is Loss Reduction Is Gain
ATP Synthase: an important enzyme that provides energy for the cell to use through the synthesis of ATP.
Glycolysis: the first step of aerobic cellular respiration in which a molecule of glucose is broken down (split in half) to produce two molecules of pyruvate.
Pyruvate: this product of glycolysis is used and synthesized by many metabolic pathways. In energy generation, it can be either converted to lactate under anaerobic conditions, or broken down to water and carbon dioxide in the presence of oxygen, generating large amounts of ATP.
Acetyl CoA formation: pyruvate is degraded and combined with coenzyme A to form acetyl coenzyme A; hydrogens are released and carbon dioxide is released. Acetyl CoA is the compound that enters the Krebs (citric acid) cycle.
Krebs Cycle: the sequence of reactions by which most living cells generate energy during the process of aerobic respiration. It consumes oxygen, producing carbon dioxide and water as waste products, and converting ADP (adenosine diphosphate) to energy rich ATP.
*also known as the citric acid cycle
Substrate level phosphorylation: takes place inside the mitochondria during glycolysis and the Krebs cycle.
Oxidative Phosphorylation: the production of ATP by ATP synthase, joining ADP to a phosphate ion in solution by condensation, using the energy from the proton gradient derived from redox reactions in the electron transport chain. Takes place in the membranes of the mitochondria during the electron transport chain.
Electron Transport Chain: a series of compounds that transfer electrons from electron donors to electron acceptors via redox reactions, and couples this electron transfer with the transfer of protons (H+ ions) across a membrane.
Energy coupling: The pairing of an energy-releasing reaction with an energy-consuming reaction; the use of an exergonic process to drive and endergonic one or the transfer of energy from catabolism to anabolism.
Chemiosmosis: The movement of ions across a selectively permeable membrane, down their electrochemical gradient. More specifically, it relates to the generation of ATP by the movement of hydrogen ions across a membrane during cellular respiration. Chemiosmosis is one of the processes by which ATP is synthesized. In eukaryotes, it takes place in the mitochondria during cellular respiration and in the chloroplasts during photosynthesis. In prokaryotes it occurs in the cell membrane.
NAD+: involved in redox reactions, carrying electrons from one reaction to another. The coenzyme is, therefore, found in two forms in cells: NAD+ is an oxidizing agent. It accepts electrons from other molecules and becomes reduced. This reaction forms NADH.
NADH: can then be used as a reducing agent to donate electrons. If oxygen is available in the system, the NADH will be oxidized readily, though indirectly, and the high-energy electrons from the hydrogen released in this process will be used to produce ATP.
FAD: a redox cofactor involved in several important reactions in metabolism. FAD can exist in two different redox states, which it converts between by accepting or donating electrons. FAD accepts two electrons and two protons to become FADH2
FADH2: reverts to FAD, sending its two high-energy electrons through the electron transport chain; the energy in FADH2 is enough to produce 1.5 equivalents of ATP by oxidative phosphorylation.
Oxidation-Reduction (redox) reactions: the gain or loss of electrons. In a reduction reaction, electrons are gained. In an oxidation reaction, electrons are lost. Use the acronym OIL RIG to remember this :Oxidation Is Loss Reduction Is Gain
ATP Synthase: an important enzyme that provides energy for the cell to use through the synthesis of ATP.
Glycolysis: the first step of aerobic cellular respiration in which a molecule of glucose is broken down (split in half) to produce two molecules of pyruvate.
Pyruvate: this product of glycolysis is used and synthesized by many metabolic pathways. In energy generation, it can be either converted to lactate under anaerobic conditions, or broken down to water and carbon dioxide in the presence of oxygen, generating large amounts of ATP.
Acetyl CoA formation: pyruvate is degraded and combined with coenzyme A to form acetyl coenzyme A; hydrogens are released and carbon dioxide is released. Acetyl CoA is the compound that enters the Krebs (citric acid) cycle.
Krebs Cycle: the sequence of reactions by which most living cells generate energy during the process of aerobic respiration. It consumes oxygen, producing carbon dioxide and water as waste products, and converting ADP (adenosine diphosphate) to energy rich ATP.
*also known as the citric acid cycle
Substrate level phosphorylation: takes place inside the mitochondria during glycolysis and the Krebs cycle.
Oxidative Phosphorylation: the production of ATP by ATP synthase, joining ADP to a phosphate ion in solution by condensation, using the energy from the proton gradient derived from redox reactions in the electron transport chain. Takes place in the membranes of the mitochondria during the electron transport chain.
Electron Transport Chain: a series of compounds that transfer electrons from electron donors to electron acceptors via redox reactions, and couples this electron transfer with the transfer of protons (H+ ions) across a membrane.
Energy coupling: The pairing of an energy-releasing reaction with an energy-consuming reaction; the use of an exergonic process to drive and endergonic one or the transfer of energy from catabolism to anabolism.
Chemiosmosis: The movement of ions across a selectively permeable membrane, down their electrochemical gradient. More specifically, it relates to the generation of ATP by the movement of hydrogen ions across a membrane during cellular respiration. Chemiosmosis is one of the processes by which ATP is synthesized. In eukaryotes, it takes place in the mitochondria during cellular respiration and in the chloroplasts during photosynthesis. In prokaryotes it occurs in the cell membrane.
NAD+: involved in redox reactions, carrying electrons from one reaction to another. The coenzyme is, therefore, found in two forms in cells: NAD+ is an oxidizing agent. It accepts electrons from other molecules and becomes reduced. This reaction forms NADH.
NADH: can then be used as a reducing agent to donate electrons. If oxygen is available in the system, the NADH will be oxidized readily, though indirectly, and the high-energy electrons from the hydrogen released in this process will be used to produce ATP.
FAD: a redox cofactor involved in several important reactions in metabolism. FAD can exist in two different redox states, which it converts between by accepting or donating electrons. FAD accepts two electrons and two protons to become FADH2
FADH2: reverts to FAD, sending its two high-energy electrons through the electron transport chain; the energy in FADH2 is enough to produce 1.5 equivalents of ATP by oxidative phosphorylation.
For a quick introduction to Cellular Respiration, check out this video! http://www.mhhe.com/biosci/bio_animations/MH01_CellularRespiration_Web/index.html